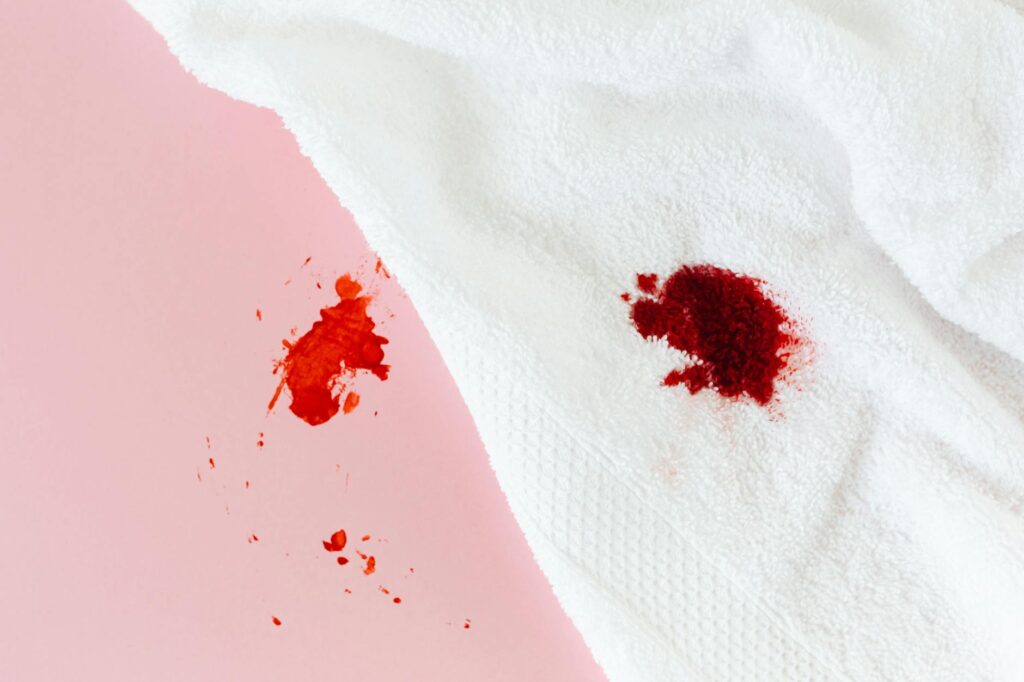
Benzoyl peroxide bleaches your towels because it is, quite literally, a peroxide — a powerful oxidizing agent that strips color from fabric on contact. The same chemical mechanism that kills acne-causing bacteria by flooding pores with oxygen also breaks apart the dye molecules woven into your towels, sheets, and pillowcases. If you have ever pulled a dark washcloth away from your face after your evening routine and noticed an orange or white splotch that was not there before, you have already seen this in action. It does not wash out, it does not fade back, and no amount of detergent will reverse it.
The bleaching is permanent. This is not a defect in your skincare product or a sign you are using too much. Even trace amounts of benzoyl peroxide — residue left on your fingers after applying a 2.5% spot treatment — can discolor fabric within minutes. The reaction begins as soon as the compound touches dye, and it accelerates when the fabric is damp. In this article, we will break down the chemistry behind why this happens, which fabrics are most vulnerable, how to protect your linens without giving up an effective acne treatment, and whether alternatives like salicylic acid carry the same risk.
Table of Contents
- How Does Benzoyl Peroxide Chemically Bleach Fabric?
- Which Fabrics and Colors Are Most Vulnerable to Bleaching?
- The Pillowcase Problem and Overnight Treatments
- How to Protect Your Towels Without Giving Up Benzoyl Peroxide
- Can You Wash Out Benzoyl Peroxide Before It Bleaches?
- Do Alternatives Like Salicylic Acid Also Bleach Fabric?
- Formulation Trends and What Is Changing
- Conclusion
- Frequently Asked Questions
How Does Benzoyl Peroxide Chemically Bleach Fabric?
Benzoyl peroxide belongs to the organic peroxide family, meaning its molecular structure contains an oxygen-oxygen bond that is inherently unstable. When it contacts organic dye molecules in fabric, that bond breaks apart and releases highly reactive oxygen radicals. These radicals attack the chromophore groups in textile dyes — the specific molecular structures responsible for absorbing light and producing color. Once the chromophore is disrupted, the dye can no longer reflect its original color, and you are left with a pale, bleached-out patch. This is the same fundamental chemistry behind hydrogen peroxide and household bleach, just delivered through a different compound. What makes benzoyl peroxide particularly aggressive toward fabric is that it does not need to be in liquid form to cause damage.
Even after your face wash has dried, a thin film of the active ingredient remains on your skin. When you press a towel against your face, the moisture from the damp fabric reactivates the compound and initiates the oxidation reaction. A clinical study published in the Journal of the American Academy of Dermatology confirmed that benzoyl peroxide residue remains active on skin for hours after application, which is partly why it works so well against bacteria — and why your pillowcases are never safe. Compared to hydrogen peroxide, which is water-soluble and rinses away relatively easily, benzoyl peroxide is lipophilic. It clings to oils on your skin and resists casual rinsing. This means a quick splash of water after applying your treatment is not enough to remove it from your hands or face before touching fabric.

Which Fabrics and Colors Are Most Vulnerable to Bleaching?
Dark and richly dyed fabrics suffer the most visible damage. Navy blue, black, burgundy, and deep green towels will show bleach marks almost immediately, often appearing as bright orange or pinkish splotches rather than white. This happens because the underlying base color of most dark-dyed cotton is warm-toned, so when the surface dye is stripped, what remains is an orange or salmon hue. White and very light-colored fabrics, on the other hand, show little to no visible change because there is minimal dye to destroy in the first place. Cotton and cotton-blend fabrics are the most susceptible because cotton fibers absorb and hold moisture, giving the benzoyl peroxide more time to react with the dye.
Synthetic fabrics like polyester and nylon use different dye-bonding processes and tend to resist bleaching somewhat better, though they are not immune. Microfiber towels, for instance, hold up longer than standard cotton terry cloth but will still eventually show discoloration with repeated exposure. However, if you are using a high-concentration benzoyl peroxide product — anything above 5% — even synthetic blends will bleach on first contact. One important limitation to note: fabric treatments marketed as “colorfast” or “fade-resistant” do not protect against peroxide bleaching. Those treatments are designed to resist fading from repeated washing and sunlight exposure, not chemical oxidation. Do not assume an expensive set of hotel-quality towels is safe just because the label makes durability claims.
The Pillowcase Problem and Overnight Treatments
overnight benzoyl peroxide treatments create the most widespread fabric damage because the compound sits against your pillowcase for six to eight hours straight. A pea-sized amount of a 2.5% benzoyl peroxide gel applied before bed is enough to leave a noticeable mark on a dark pillowcase by morning. Dermatologists frequently recommend nighttime application because the extended contact time improves efficacy against acne, but they rarely mention the linen cost that comes with it. The pattern is predictable: you will see the first bleach marks on the area of the pillowcase where your face rests, then gradually on the edges as you shift during sleep. Over a few weeks, what started as a small spot becomes an irregular, mottled pattern across the entire surface.
Many people do not connect the discoloration to their acne treatment right away and instead blame their detergent or washing machine. One tell-tale sign it is benzoyl peroxide and not laundry damage is the color of the bleach mark — if dark fabric turns orange or coral rather than white, a peroxide is almost certainly responsible. Sheets are vulnerable too, particularly if you sleep without a shirt. Benzoyl peroxide body washes and back-acne sprays will bleach fitted sheets, duvet covers, and even mattress protectors. If you use a body wash containing benzoyl peroxide in the shower and do not rinse thoroughly, the residue transfers to your towel and then to whatever the towel touches next.

How to Protect Your Towels Without Giving Up Benzoyl Peroxide
The most reliable solution is also the simplest: dedicate specific towels and pillowcases to your acne routine and accept that they will get bleached. Buy inexpensive white or light-colored towels exclusively for face use. White cotton is effectively immune to visible bleaching because there is no dye to strip. This approach costs far less over time than repeatedly replacing your good towels. For pillowcases, you have two practical options. The first is the white pillowcase strategy — keep a rotation of cheap white cotton pillowcases and swap them every two to three nights, which is good hygiene practice regardless.
The second is to apply your benzoyl peroxide treatment earlier in the evening, wait 20 to 30 minutes for it to fully absorb, then gently blot your face with a damp white cloth to remove surface residue before going to bed. This reduces but does not eliminate transfer. The tradeoff is real: removing excess product from your skin means slightly less active ingredient working overnight, which may marginally reduce the treatment’s effectiveness. For most people with mild to moderate acne, this reduction is negligible. For severe cystic acne where every percentage point of concentration matters, talk to your dermatologist before altering your application routine. Some people try applying benzoyl peroxide only in the morning to spare their bedding, but this introduces a different problem — the compound increases photosensitivity, and daytime use requires diligent sunscreen application on top of it.
Can You Wash Out Benzoyl Peroxide Before It Bleaches?
No — and this is the most common misconception. By the time you see the stain, the dye has already been chemically destroyed. The bleach mark is not a deposit sitting on top of the fabric. It is the absence of color where dye molecules have been broken apart. No stain remover, vinegar soak, or enzymatic cleaner will restore the original color because there is nothing to remove or reactivate. The dye is gone.
Washing your towels immediately after use does help prevent further spread of the benzoyl peroxide residue to other items in the hamper or washing machine. If a towel with wet benzoyl peroxide residue sits in a laundry basket pressed against a dark shirt, that shirt will bleach too. This cross-contamination is an underappreciated hazard. Keep used face towels separate from the rest of your laundry, or rinse them under water before tossing them in the hamper. One warning worth repeating: do not attempt to re-dye bleached towels as a fix. The oxidized areas will not hold dye the same way as the surrounding fabric, and you will end up with an uneven, patchy result that looks worse than the original bleach mark.

Do Alternatives Like Salicylic Acid Also Bleach Fabric?
Salicylic acid does not bleach towels. It is a beta-hydroxy acid, not an oxidizer, so it lacks the reactive oxygen mechanism that destroys dye. If fabric staining is a dealbreaker for you, salicylic acid-based cleansers and treatments are a safe alternative from a textile perspective. Adapalene (Differin), another popular over-the-counter acne treatment, also does not cause bleaching.
Neither does azelaic acid or niacinamide. However, switching away from benzoyl peroxide purely to save your towels means giving up one of the most effective topical antibacterial agents available for acne. Salicylic acid excels at exfoliation and pore-clearing but does not kill Cutibacterium acnes bacteria the way benzoyl peroxide does. For inflammatory acne with red, swollen papules and pustules, benzoyl peroxide is often the better choice clinically. The decision comes down to whether you would rather manage your laundry or manage potentially less effective treatment.
Formulation Trends and What Is Changing
Pharmaceutical companies are aware of the fabric-staining complaint, and some newer formulations are designed to minimize transfer. Encapsulated benzoyl peroxide products use a slow-release delivery system that keeps more of the active ingredient on the skin and less on the surface where it can rub off onto fabric.
These formulations are not stain-proof — the compound is still benzoyl peroxide — but early user reports suggest noticeably less transfer to pillowcases compared to traditional gels and creams. There is also growing interest in benzoyl peroxide combined with moisturizing bases that help the product absorb faster, reducing the window during which wet residue can contact fabric. As these formulations mature, the towel-bleaching problem may become less severe, though it is unlikely to disappear entirely as long as the active ingredient remains a peroxide.
Conclusion
Benzoyl peroxide bleaches your towels because it is an oxidizing agent that irreversibly destroys fabric dye on contact. No washing technique, fabric treatment, or laundry hack will prevent or reverse this. The chemistry is non-negotiable.
Dark fabrics are hit hardest, cotton is more vulnerable than synthetics, and overnight treatments cause the most widespread damage to bedding. The practical path forward is straightforward: use dedicated white towels and pillowcases for your acne routine, keep benzoyl peroxide-contaminated laundry separate from other clothes, and do not waste money trying to restore bleached fabric. If you want to reduce transfer to bedding, apply your treatment earlier in the evening and blot away excess before sleeping. And if the staining truly bothers you more than your acne does, talk to your dermatologist about whether a non-peroxide alternative might work for your skin.
Frequently Asked Questions
How long does benzoyl peroxide take to bleach a towel?
Contact as brief as 30 seconds can cause visible discoloration on dark fabric. The reaction begins immediately, though a faint mark may take a few minutes to become obvious to the eye.
Will a lower concentration of benzoyl peroxide cause less bleaching?
Lower concentrations (2.5%) cause slightly less dramatic bleaching than higher ones (10%), but all concentrations will bleach fabric. Even the lowest available strength is a peroxide and will strip dye.
Does rinsing my face before using a towel prevent bleaching?
Rinsing helps reduce the amount of active residue, but it does not eliminate it. Benzoyl peroxide is oil-soluble and clings to skin even after splashing water on your face. You would need a thorough cleanse with a non-medicated face wash to meaningfully reduce transfer.
Can I use a barrier cream on my face to stop the benzoyl peroxide from touching my pillow?
Applying a heavy moisturizer or occlusive like Vaseline over your benzoyl peroxide treatment can reduce some surface transfer, but it also potentially interferes with how the medication works. This is not a widely recommended approach.
Are there any towel materials that resist benzoyl peroxide bleaching?
White towels of any material are effectively immune to visible damage. Among colored options, dark polyester blends resist slightly better than cotton but will still bleach with repeated exposure. No commercially available dyed fabric is truly peroxide-proof.
You Might Also Like
- What Causes Acne When You Start Adapalene
- How to Tell If You’re Purging or Breaking Out from a New Product
- Why Retinol Makes Acne Worse Before It Gets Better
Browse more: Acne | Acne Scars | Adults | Back | Blackheads