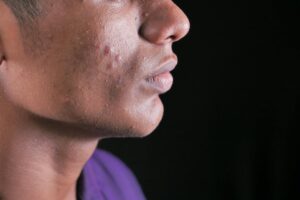
Leaky gut shows up on your skin as persistent, seemingly unexplainable inflammation — think chronic acne that doesn’t respond to topical treatments, rosacea flares that worsen after meals, eczema patches that migrate around your body, or a dull, reactive complexion that never quite calms down. These aren’t random skin problems. They’re visible signals that your intestinal barrier has become too permeable, allowing partially digested food proteins, bacterial toxins called lipopolysaccharides, and other inflammatory molecules to slip into your bloodstream and trigger immune responses that manifest on your largest organ. A person might spend years cycling through retinoids, antibiotics, and expensive serums when the real issue is happening several feet south of their face.
The connection between gut permeability and skin disease isn’t new or fringe. Dermatologists in the 1930s proposed the gut-skin axis, and modern research has confirmed that patients with acne, rosacea, and atopic dermatitis frequently show markers of increased intestinal permeability. One 2008 study in the journal Gut Pathogens found that small intestinal bacterial overgrowth — a condition closely tied to leaky gut — was ten times more prevalent in rosacea patients than in controls. This article breaks down exactly which skin symptoms point to a compromised gut lining, how the inflammatory cascade works from intestine to epidermis, which conditions are most commonly linked, and what actually helps versus what is just wellness marketing noise.
Table of Contents
- How Does a Leaky Gut Actually Cause Skin Problems?
- The Specific Skin Conditions Linked to Intestinal Permeability
- Food Reactions That Show Up on Your Face
- Testing and Identifying Gut-Driven Skin Issues
- Why Standard Acne Treatments Fail When the Gut Is Involved
- What Actually Helps Repair the Gut-Skin Connection
- The Evolving Science of the Gut-Skin Axis
- Conclusion
- Frequently Asked Questions
How Does a Leaky Gut Actually Cause Skin Problems?
Your intestinal lining is a single-cell-thick barrier held together by tight junction proteins like claudin, occludin, and zonulin. When functioning properly, this barrier selectively absorbs nutrients while keeping bacteria, undigested food particles, and toxins confined to the gut lumen. When tight junctions loosen — due to chronic stress, alcohol, NSAIDs, dysbiosis, or dietary triggers like excessive gluten in sensitive individuals — the barrier becomes permeable. Molecules that should never enter the bloodstream start leaking through, and your immune system treats them as invaders. The result is a systemic inflammatory response that doesn’t stay contained to the gut. The skin gets hit because it’s densely packed with immune cells and blood vessels that carry these circulating inflammatory mediators everywhere. When lipopolysaccharides from gram-negative gut bacteria enter the bloodstream (a process called endotoxemia), they activate toll-like receptors on immune cells throughout the body, including in the dermis.
This triggers production of inflammatory cytokines — IL-6, TNF-alpha, IL-1 beta — that increase sebum production, disrupt skin barrier function, and accelerate keratinocyte turnover in ways that clog pores. Compare this to a standard hormonal acne pattern, where breakouts follow a predictable monthly cycle and concentrate around the jawline. Gut-driven skin inflammation tends to be more diffuse, less cyclical, and stubbornly resistant to standard dermatological treatments. The gut-skin connection also works through the microbiome directly. Your gut bacteria produce short-chain fatty acids, vitamins, and neurotransmitters that regulate skin homeostasis. When dysbiosis disrupts these outputs, the skin loses access to compounds it depends on for proper barrier function and immune regulation. This is why someone with leaky gut might notice not just acne, but also increased skin sensitivity, slower wound healing, and a general loss of resilience in their complexion.

The Specific Skin Conditions Linked to Intestinal Permeability
The strongest research connections exist between leaky gut and four skin conditions: acne vulgaris, rosacea, atopic dermatitis (eczema), and psoriasis. Each presents differently, but they share a common upstream trigger in gut-derived systemic inflammation. Acne patients with concurrent gut issues often have inflammatory papules and cysts that appear across the cheeks, forehead, and temples rather than the classic hormonal pattern along the jawline. Their skin is frequently oily yet dehydrated — a paradox that makes more sense when you understand that systemic inflammation disrupts both sebum regulation and the skin’s moisture barrier simultaneously. Rosacea has one of the tightest links to gut permeability. The condition’s hallmark — facial flushing, visible blood vessels, and inflammatory papules concentrated on the central face — worsens significantly when the gut is compromised.
Research published in the British Journal of Dermatology demonstrated that treating small intestinal bacterial overgrowth in rosacea patients led to complete remission in many cases, a result that topical treatments rarely achieve. Eczema, particularly in adults who develop it later in life or whose childhood eczema returns after years of remission, frequently correlates with gut permeability changes triggered by stress, dietary shifts, or antibiotic use. However, not every case of these conditions traces back to the gut. If your acne is clearly cyclical and responds well to topical retinoids, or if your eczema is limited to areas of skin contact with a known allergen, leaky gut is probably not your primary driver. The gut-skin axis matters most when skin conditions are widespread, resistant to standard treatment, associated with digestive symptoms, or worsened predictably by certain foods. Jumping to gut-healing protocols without this context can lead to unnecessary restriction and expense while the actual trigger — a contact allergy, hormonal imbalance, or medication side effect — goes unaddressed.
Food Reactions That Show Up on Your Face
One of the most telling signs of leaky gut is developing skin reactions to foods that never bothered you before. This happens because increased intestinal permeability allows larger protein molecules to enter the bloodstream intact, where the immune system flags them as foreign. Over time, you develop IgG-mediated sensitivities — not true allergies, but delayed hypersensitivity reactions — that can manifest as facial flushing, under-the-skin cystic bumps, perioral dermatitis, or generalized puffiness that appears 12 to 72 hours after eating the trigger food. The delay makes it incredibly difficult to identify the cause without careful tracking. A common example: someone starts breaking out in deep, painful cysts along their cheeks and jawline every few days. They’ve tried benzoyl peroxide, salicylic acid, prescription antibiotics, even Accutane, with limited improvement.
After keeping a detailed food and skin diary for six weeks, they notice that flares consistently follow meals heavy in dairy and gluten — foods they ate without issue for decades. This pattern points strongly toward increased intestinal permeability rather than a lifelong allergy. The foods themselves aren’t inherently problematic; the broken barrier is allowing them to provoke an immune response they previously couldn’t. The tricky part is that elimination diets, while diagnostically useful, don’t fix the underlying permeability issue. Removing dairy and gluten might calm the skin temporarily, but if the gut lining remains compromised, new sensitivities often develop to whatever replacement foods become dietary staples. People who restrict extensively without addressing the gut itself sometimes end up reactive to dozens of foods, which creates nutritional deficiencies that further worsen skin health. The goal should be identifying and removing the most inflammatory triggers while simultaneously working to restore the intestinal barrier, not permanent restriction.

Testing and Identifying Gut-Driven Skin Issues
If you suspect your skin problems trace back to your gut, there are a few diagnostic pathways worth considering, though none of them are perfect. The lactulose-mannitol test is the most direct measure of intestinal permeability — you drink a solution containing two sugars of different molecular sizes, and urine collection measures how much of each passes through. Elevated lactulose in urine indicates that the barrier is letting through molecules it shouldn’t. This test is validated in research settings but not widely available in standard dermatology or primary care practices, and insurance coverage is inconsistent. More practically accessible are stool tests like comprehensive digestive stool analysis, which measures markers of inflammation (calprotectin, secretory IgA), digestion efficiency, and microbial diversity.
Zonulin blood testing, which measures a protein that regulates tight junction permeability, has gained popularity in functional medicine circles but has significant accuracy limitations — commercial zonulin assays often measure related proteins rather than zonulin itself, leading to false positives. Compared to these lab approaches, a well-executed elimination diet with systematic reintroduction remains one of the most informative tools, costing nothing and providing personalized data that no blood panel can replicate. The tradeoff is time and discipline. Lab testing gives you data points in days but may miss the full picture or provide misleading specificity. An elimination protocol takes four to eight weeks of strict adherence followed by methodical reintroduction, but tells you exactly which foods provoke your particular skin symptoms. For most people dealing with treatment-resistant skin conditions, combining a basic inflammation panel (CRP, fecal calprotectin) with a 30-day elimination of the most common gut irritants — gluten, dairy, alcohol, refined sugar, and processed seed oils — provides the clearest signal with the least expense.
Why Standard Acne Treatments Fail When the Gut Is Involved
The frustrating cycle of gut-driven skin problems is that conventional treatments address the downstream symptoms while ignoring the upstream cause. Topical retinoids increase cell turnover and reduce comedones, but they do nothing about the systemic inflammation driving excess sebum production and immune-mediated breakouts. Oral antibiotics like doxycycline can actually worsen the situation over time — they may temporarily reduce skin bacteria and inflammation, but they simultaneously disrupt the gut microbiome, potentially increasing intestinal permeability and creating a rebound effect once the course ends. Even isotretinoin (Accutane), the most powerful acne medication available, has limitations when gut permeability is a significant contributing factor. While it dramatically reduces sebum production and can induce long-term remission for many patients, those with underlying gut issues sometimes experience relapse within months of completing treatment.
There’s also evidence, though not yet conclusive, that isotretinoin itself may affect gut barrier function — case reports of inflammatory bowel disease onset during or after isotretinoin treatment have appeared in the literature, though large epidemiological studies haven’t confirmed a causal link. The point isn’t to avoid these treatments entirely but to recognize that if standard approaches keep failing or effects don’t last, the gut deserves investigation. Spironolactone for hormonal acne presents a similar limitation. It works by blocking androgen receptors, which is effective when hormonal fluctuation is the primary driver. But when gut-derived inflammation is amplifying hormonal sensitivity in the skin — which research suggests it can — spironolactone manages the symptom without resolving why the skin became so reactive to normal hormone levels in the first place. Layering gut repair underneath conventional treatment often produces results that neither approach achieves alone.

What Actually Helps Repair the Gut-Skin Connection
The evidence-supported approach to restoring intestinal permeability involves three concurrent strategies: removing ongoing damage, providing barrier-supportive nutrients, and rebuilding microbial diversity. Removing damage means identifying and eliminating the factors maintaining permeability — for many people, this includes chronic NSAID use, excessive alcohol, unmanaged psychological stress, and specific food triggers identified through elimination testing. A registered nurse in her 40s who had battled adult-onset cystic acne for three years found that stopping daily ibuprofen for chronic headaches (replacing it with magnesium supplementation and physical therapy) reduced her inflammatory breakouts by roughly 60 percent before she changed anything about her skincare routine.
Barrier-supportive nutrients with meaningful research behind them include L-glutamine (the primary fuel source for enterocytes that line the intestinal wall), zinc carnosine (shown to stabilize gut mucosa in multiple trials), omega-3 fatty acids (which reduce intestinal inflammation), and butyrate (a short-chain fatty acid produced by beneficial bacteria that strengthens tight junctions). Probiotic supplementation is more nuanced — specific strains like Lactobacillus rhamnosus GG and Saccharomyces boulardii have evidence for gut barrier support, while many commercial probiotics contain strains chosen for shelf stability rather than therapeutic effect. Fermented foods like sauerkraut, kimchi, and plain kefir provide both live bacteria and postbiotic compounds, though people with histamine sensitivity may find these temporarily worsen flushing and eczema until underlying gut healing progresses.
The Evolving Science of the Gut-Skin Axis
Research on the gut-skin axis is accelerating, with several promising directions. Postbiotic therapy — using the metabolic byproducts of beneficial bacteria rather than live organisms — is showing potential for people who react poorly to probiotics themselves. Fecal microbiota studies are mapping specific bacterial signatures associated with acne, rosacea, and eczema, which could eventually lead to targeted microbial interventions rather than the broad-spectrum approach of current probiotics.
Oral and topical synbiotic products that pair specific prebiotic fibers with matched probiotic strains are already entering the market, though rigorous clinical trials are still catching up to commercial enthusiasm. The most important shift is the growing recognition among dermatologists that skin conditions exist within a systemic context. The era of treating the skin as an isolated organ is giving way to a more integrated understanding where gut health, stress physiology, and immune regulation are considered standard parts of the dermatological workup — particularly for patients who’ve been through multiple treatment failures. For anyone whose skin has resisted everything thrown at it, investigating what’s happening below the surface, literally, might be the missing piece.
Conclusion
Leaky gut leaves visible fingerprints on the skin — persistent inflammatory acne, rosacea that worsens after eating, eczema that wanders and resists topical treatment, and a reactive complexion that never fully stabilizes. These patterns make more sense when you understand that a compromised intestinal barrier allows inflammatory molecules into the bloodstream, triggering immune responses that play out across the skin’s surface. The key diagnostic clue is treatment resistance: when standard dermatological approaches provide incomplete or temporary relief, the gut becomes a logical place to investigate.
Addressing gut-driven skin problems requires working on both ends simultaneously. Continue appropriate topical and dermatological care for symptom management while removing gut-damaging inputs, supporting intestinal barrier repair with targeted nutrients, and rebuilding microbial diversity through diet and evidence-based supplementation. Track your food intake alongside skin symptoms for at least a month to identify patterns a dermatologist’s office visit can’t reveal. This isn’t an overnight fix — meaningful gut repair typically takes three to six months of consistent effort — but for the many people whose skin problems originate below the surface, it’s the intervention that finally makes the difference.
Frequently Asked Questions
Can leaky gut cause acne in people who never had it as a teenager?
Yes. Adult-onset acne, particularly inflammatory cystic acne appearing in your 30s or 40s, is one of the most common skin presentations of increased intestinal permeability. Gut barrier function can degrade over time due to cumulative stress, medication use, dietary changes, or microbiome shifts, triggering skin inflammation in people with no adolescent acne history.
How long does it take for skin to clear after you start healing leaky gut?
Most people report initial improvement in skin reactivity and redness within four to six weeks of consistent gut-supportive changes. Full resolution of inflammatory acne or eczema typically takes three to six months, since intestinal epithelial cells regenerate on a three-to-five-day cycle but the immune recalibration and microbiome restoration that produce lasting skin changes require much longer.
Does leaky gut cause dark circles under the eyes?
Periorbital hyperpigmentation — dark under-eye circles not explained by sleep deprivation or genetics — is frequently reported alongside gut permeability issues. The mechanism likely involves chronic low-grade inflammation causing vasodilation and increased vascular permeability in the thin under-eye skin, along with potential nutrient malabsorption (iron, B12) that contributes to the discoloration.
Can probiotics make skin worse before it gets better?
Yes, and this is common enough to have a name in functional medicine circles: a die-off or Herxheimer-like reaction. Introducing beneficial bacteria can temporarily increase skin breakouts, gas, or bloating as the microbial balance shifts. If worsening persists beyond two to three weeks or is severe, reduce the probiotic dose or switch strains rather than pushing through, as prolonged worsening may indicate histamine intolerance or a mismatch between the probiotic and your specific gut environment.
Is there a specific type of acne that points to leaky gut versus hormonal acne?
Gut-driven acne tends to be more diffuse across the face (cheeks, forehead, temples) rather than concentrated along the jawline and chin like classic hormonal acne. It also often presents with concurrent skin issues — acne plus rosacea-like flushing, or acne plus eczema patches — which suggests systemic inflammation rather than a localized hormonal trigger. Worsening after specific meals rather than around menstrual cycles is another distinguishing pattern.
You Might Also Like
- Why the IUD Caused Your Skin to Break Out
- Why Thyroid Problems Show Up on Your Skin First
- What SIBO Has to Do with Skin Breakouts
Browse more: Acne | Acne Scars | Adults | Back | Blackheads