Bioprinting technology represents one of the most promising frontiers in regenerative medicine, yet when it comes to treating acne scars, the timeline is far longer than headlines suggest. The excitement is understandable: imagine a custom-printed patch of your own skin cells that could replace scarred tissue with pristine, functional skin. But the gap between that vision and clinical reality is immense. This article examines the current state of bioprinting research, the specific technical hurdles that must be overcome, and why acne scar repair—despite being less complex than some other bioprinting applications—remains a multi-decade away problem.
Table of Contents
- Where Is Bioprinting Actually Right Now?
- Why Vascularization Remains the Core Problem
- The Missing Skin Appendages That Make Skin Actual Skin
- From Animal Models to Human Trials: The Translation Barrier
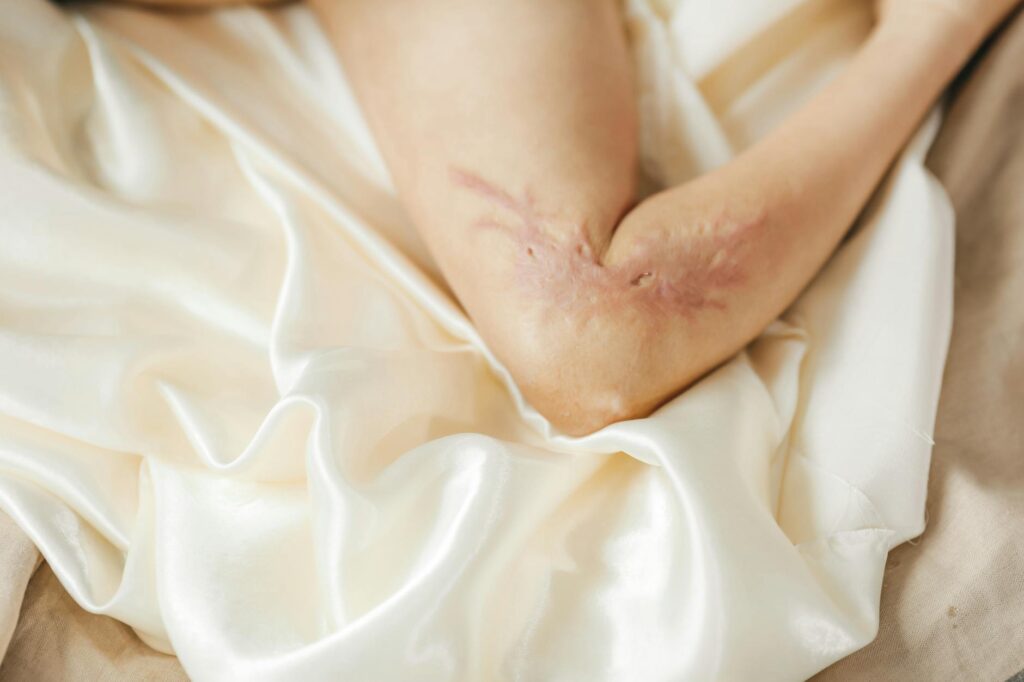
- Why Acne Scars Are Particularly Complex
- What Current Research Actually Proves (and Doesn’t)
- The Timeline and What to Realistically Expect
- What About Alternative Approaches Happening Now?
- Conclusion
- Frequently Asked Questions
Where Is Bioprinting Actually Right Now?
The current state of bioprinting for skin is still firmly in the preclinical stage. A 2025 scoping review published in a peer-reviewed journal searching clinical and observational trials involving bioprinting found that no human clinical trials of bioprinted skin have been conducted yet. Everything we know comes from laboratory research using animal models, particularly mice and rabbits. In July 2023, researchers published results from a preclinical study using bioprinted skin substitutes made from patients’ own fibroblasts and keratinocytes—the two main cell types that comprise skin—and the early results were genuinely encouraging. In mouse models with full-thickness skin defects, complete wound healing with minimal scar tissue occurred within just 14 days.
That’s remarkable progress for a technology that’s still experimental, and it explains why dermatologists and patients have started asking when this might become available for human use. However, the gap between a mouse study and human treatment is enormous. Mice heal differently than humans; their skin is thinner, their immune systems respond differently to foreign materials, and they don’t develop the kind of hypertrophic or atrophic scarring that characterizes acne damage. Moving toward humans, Poietis and Assistance Publique–Hôpitaux de Marseille announced a collaboration to conduct the first Phase I clinical trial of bioprinted skin, expected to begin within two years of the announcement. But this trial is for wound healing in general—not acne scars specifically. The distinction matters: a burn or surgical wound is a different problem than acne scarring, which involves structural damage to skin that has already healed (albeit poorly) and involves damage to hair follicles and oil gland structures.

Why Vascularization Remains the Core Problem
The biggest single obstacle to functional bioprinted skin is vascularization—the ability to create working blood vessels within the printed tissue. Your skin is alive because it has a rich network of blood vessels delivering oxygen and nutrients. Bioprinted skin currently can’t replicate this. Researchers have managed to print single-cell-layer tubes that resemble blood vessels, but creating the complex, multiscale vascular architecture that real skin requires remains, in the words of recent peer-reviewed research, “still a great challenge” with “a long way to go.” The fundamental problem is that printed structures larger than about 200 microns thick begin to die from the center outward because nutrients can’t diffuse far enough to keep the cells alive.
For acne scar repair specifically, this is particularly limiting. A severely scarred area of skin doesn’t just need a cosmetic cover—it needs functional integration with the surrounding tissue, full blood supply, and the ability to respond to temperature changes and support healing. Without vascularization, bioprinted skin is essentially a non-living patch that might fill a hole visually but won’t behave like real skin. Some researchers are experimenting with printing pre-vascularized structures and then implanting them with the hope that the body’s own blood vessels will connect to them, but this remains largely theoretical and hasn’t been demonstrated reliably in human tissue.
The Missing Skin Appendages That Make Skin Actual Skin
Beyond blood vessels, there’s another critical limitation: bioprinted constructs currently lack hair follicles and sebaceous glands (oil glands). This might seem like a cosmetic detail, but it’s fundamental to skin function and, importantly, it’s directly relevant to acne scarring. Acne happens because hair follicles and sebaceous glands become inflamed and infected; when severe acne heals, it often leaves scarring precisely in these follicular structures. A 2024 research review noted that obtaining hair follicles and sebaceous glands in bioprinted constructs “still seems far away.” Without these structures, bioprinted skin might look smoother under the surface, but it won’t truly replace the damaged acne-scarred area.
It would be like having skin that can’t sweat properly, can’t produce the oils that protect skin from drying out, and can’t grow hair—in other words, functionally deficient skin. Researchers are exploring methods to co-print multiple cell types and growth factors that might promote the formation of these appendages, but the complexity is enormous. Hair follicles are intricate, three-dimensional structures that require interaction between different cell types, precise anatomical orientation, and specific signaling molecules to develop. Getting this right in a bioprinter—where you’re depositing material layer by layer, sometimes just microns thick—is exponentially harder than it sounds.

From Animal Models to Human Trials: The Translation Barrier
The jump from printing skin that works in a petri dish or a mouse to printing skin that works in a human being involves far more than just scale. There are regulatory barriers, manufacturing challenges, questions about cell sourcing and preservation, transportation logistics, cost, and ethical concerns in cell therapy. The FDA and equivalent agencies in other countries don’t just want to see that something works in mice; they want robust evidence of safety, manufacturing consistency, long-term outcomes, and clear protocols for what happens if something goes wrong. The upcoming Poietis trial is Phase I, which means it’s primarily about safety and tolerability—proving that the technology doesn’t cause harm—rather than proving that it actually repairs acne scars.
Manufacturing at scale presents another hurdle entirely. A bioprinter that can produce a small patch of skin suitable for a mouse study or even a single human patient is very different from a facility that can reliably produce dozens or hundreds of individualized skin patches on a weekly basis. Each patient’s bioprinted skin would ideally be made from their own cells, which means the process needs to be customized, repeatable, and fast enough to treat patients in a reasonable timeframe. If it takes three months to grow cells and print skin for one patient, that’s not a scalable treatment. Add in the challenge of preserving and transporting living tissue—bioprinted skin can’t just be frozen and shipped like a drug—and the logistical complexity becomes apparent.
Why Acne Scars Are Particularly Complex
Acne scarring presents unique challenges that distinguish it from the general wound-healing applications that bioprinting research has focused on. Acne scars come in several types—atrophic (depressed), hypertrophic (raised), and sometimes a combination—and they result from the destruction of collagen in deeper skin layers, often with damage to the follicular structures themselves. When bioprinting researchers successfully healed full-thickness wounds in mouse models, those were fresh injuries with clean tissue planes. An acne scar is old, remodeled tissue with altered collagen architecture and compromised structural integrity.
Printing new skin on top of or into scar tissue requires not just depositing new cells, but potentially remodeling or replacing the underlying scar tissue itself—a far more complex problem. Additionally, acne scars often occur in areas of the face that are highly visible and cosmetically sensitive. Even if a bioprinted skin patch functionally works, patients will expect it to match surrounding skin in texture, tone, and appearance. This level of aesthetic precision is beyond current bioprinting capabilities, which struggle with color consistency and surface texture control. A bioprinted patch that’s functionally sound but obviously different in appearance from surrounding skin might not be considered successful from a patient’s perspective.

What Current Research Actually Proves (and Doesn’t)
The 2023 mouse study demonstrating 14-day wound closure is real progress, but it’s important to understand exactly what it demonstrated and what it didn’t. The study showed that bioprinted skin constructs made from a patient’s own cells could integrate into a wound site and promote healing without causing immune rejection or infection. This is genuinely important—it proves the concept that bioprinted skin can function within a living organism, at least in a small animal model.
The use of autologous cells (the patient’s own cells) is also significant because it sidesteps immune rejection, one of the major barriers to transplantation. However, this study didn’t demonstrate long-term durability, didn’t test on acne-scarred tissue, didn’t incorporate blood vessels or skin appendages, and didn’t prove that the healed tissue was truly indistinguishable from normal skin on histological examination. It’s a proof-of-concept that the broad approach—bioprinting cells into a scaffold and implanting it—can work, but it’s still a very far distance from “ready for acne scar patients.”.
The Timeline and What to Realistically Expect
Based on current announcements and the state of research, here’s what the realistic timeline looks like. Within roughly two years from now (around 2028), the first Phase I human clinical trial of bioprinted skin should begin, focusing on wound healing. This trial will primarily assess safety. If it goes well, Phase II trials might begin around 2030-2032, examining whether it actually works better than current standard treatments. Even if Phase II is successful, Phase III trials with larger patient populations would likely follow, potentially extending into the mid-2030s.
Only after successful Phase III trials would the FDA or equivalent agencies consider approval. This timeline is for wound healing, not acne scars. Specific trials for acne scarring would likely begin after general wound-healing applications are established, adding another 5-10 years. And this is assuming no major setbacks, no manufacturing challenges that force redesigns, and no unexpected safety signals. In reality, bioprinting for acne scars—specifically, as a mainstream clinical treatment—is likely 15-25 years away, not 5-10. That puts us realistically in the 2040s or 2050s for widespread availability, which is genuinely decades.
What About Alternative Approaches Happening Now?
While waiting for bioprinting to mature, the dermatology field hasn’t been idle. Patients with acne scars today have options ranging from topical treatments and chemical peels to microneedling, laser resurfacing, subcision, dermal fillers, and even fat grafting or skin grafting from other areas of the body. These treatments work reasonably well for many patients and have decades of safety data. Laser resurfacing, for example, can provide meaningful improvement in atrophic acne scars by ablating damaged tissue and stimulating collagen remodeling.
It’s not perfect, and results vary based on scar type and skin type, but it’s available today. This creates a practical dilemma for patients: you can have a moderately effective treatment that works now, or you can wait potentially 20+ years for a theoretically superior treatment that might eventually become available. For someone in their 20s or 30s with significant acne scarring, the psychological and social impact of waiting decades is substantial. It’s worth understanding that bioprinting isn’t competing against nothing; it’s competing against treatments that, while imperfect, actually exist and can provide real improvement in scar appearance and sometimes function.
Conclusion
Bioprinting skin for acne scar repair remains decades away primarily because the technology hasn’t progressed to human testing yet, and the obstacles between preclinical success in mice and clinical utility in humans are formidable. Vascularization, skin appendages, manufacturing scale, regulatory approval, and the unique complexity of acne scarring all represent multi-year or multi-decade challenges. The first human clinical trial will likely focus on wound healing rather than acne scars, and even that is still years away.
While recent preclinical results are genuinely encouraging—showing that bioprinted skin can integrate into living tissue and promote healing—there’s an enormous gap between a mouse healing completely in 14 days and a human patient with acne scars receiving a custom-printed skin patch that looks perfect and functions normally. For anyone currently dealing with acne scars, the practical takeaway is clear: established treatments like laser resurfacing, microneedling, and other modalities offer real improvements today, while bioprinting remains a future technology that may eventually exceed current approaches but shouldn’t factor into treatment decisions for the foreseeable future. Following the development of bioprinting research is reasonable for understanding where dermatology is heading, but patients seeking scar improvement should focus on the effective options available now rather than waiting for a technology that’s likely still 15-25 years from becoming a mainstream treatment option.
Frequently Asked Questions
How much longer until bioprinted skin is available for treating acne scars?
Based on current development timelines, the first human clinical trials of bioprinted skin should begin around 2028, but those will focus on wound healing, not acne scars. Acne-specific trials would likely follow several years later. Realistic widespread availability for acne scar treatment is estimated at 15-25 years, placing it in the 2040s-2050s at the earliest.
Why can’t they just use the mouse results to treat patients now?
Animal studies, while promising, don’t directly translate to humans. Mice have different skin structure, healing responses, and immune systems than humans. The FDA and international regulatory bodies require human clinical trials demonstrating safety and effectiveness before approving new medical treatments. There are no shortcuts through this process for patient safety reasons.
Will bioprinted skin be better than current acne scar treatments?
Potentially, yes—if it eventually works as intended. Bioprinted skin made from your own cells could theoretically provide complete integration, natural appearance, and normal skin function (including blood supply and skin appendages). However, current treatments like laser resurfacing and microneedling already provide meaningful improvement for many patients and are available today.
What’s the biggest technical challenge holding bioprinting back for skin?
Vascularization—creating functional blood vessels within the printed tissue. Current bioprinting can only create very thin structures with single-cell-layer tubes that don’t work like real blood vessels. Without solving this, bioprinted skin patches lack nutrient delivery and won’t truly integrate with surrounding tissue.
Could bioprinting solve all types of acne scars?
Theoretically it might help, but acne scars come in different types (atrophic, hypertrophic, ice-pick scars), and each would present different challenges. Additionally, deep scars involving damage to hair follicles and oil glands would be particularly complex because bioprinted skin currently can’t reliably recreate these structures.
If I have acne scars now, should I wait for bioprinting or get treatment today?
For anyone currently dealing with significant acne scarring, established treatments available today—laser resurfacing, microneedling, subcision, dermal fillers—can provide real improvement. Waiting 15-25 years for a technology that may or may not become mainstream is not practical. Discuss treatment options with a dermatologist rather than waiting on bioprinting.
You Might Also Like
- What Naltrexone Low-Dose Does for Inflammatory Skin and Acne
- What i-PRF vs L-PRF Means for Acne Scar Treatment Protocols
- What XoGlo Exosomes Do for Acne Scar Remodeling
Browse more: Acne | Acne Scars | Adults | Back | Blackheads