LaViv (Azficel-T) received FDA approval on June 21, 2011, as the first and only personalized aesthetic cell therapy approved specifically for moderate to severe nasolabial fold wrinkles—those deep smile lines that run from the nose to the corners of the mouth. The FDA granted this approval based on robust clinical evidence from two Phase III randomized, double-blind, placebo-controlled trials involving 421 patients, which demonstrated that the treatment could achieve a two-point improvement in wrinkle severity at six months post-treatment. Unlike many aesthetics procedures that rely on temporary fillers or neurotoxins, LaViv works by using your own skin cells, making it a fundamentally different approach to facial rejuvenation that appealed to the FDA’s regulatory framework for cellular therapies.
However, it’s critical to understand that while the title mentions scars, LaViv is not FDA-approved for acne scarring or other scar types, despite research exploring its potential in that area. The FDA’s approval was narrowly focused on nasolabial folds because that’s where the clinical trial data demonstrated safety and efficacy. This article explains why LaViv succeeded in gaining approval for this specific indication, how it works, what the clinical evidence showed, and why the scar application remains off-label despite preliminary interest.
Table of Contents
- Why the FDA Approved LaViv for Nasolabial Folds but Not Other Indications
- The Clinical Trial Design That Led to FDA Approval
- How LaViv Differs from Dermal Fillers and Botox for Nasolabial Folds
- The LaViv Treatment Process: From Biopsy to Injection
- Why LaViv Wasn’t Approved for Acne Scars Despite Research Potential
- Clinical Outcomes and Safety Data from the Pivotal Trials
- The Future of Cellular Therapies in Aesthetics and Where LaViv Stands
- Conclusion
Why the FDA Approved LaViv for Nasolabial Folds but Not Other Indications
The FDA’s decision to approve LaViv for nasolabial folds stemmed directly from the quality and design of the clinical evidence presented. Two identical Phase III multi-center studies enrolled 421 patients total and followed a rigorous, blinded protocol where neither the patients nor the evaluators knew who received treatment versus placebo. The studies measured a clear, objective primary endpoint: a two-point improvement on a standardized wrinkle severity scale at six months following the third treatment session, with injections spaced approximately 5 weeks apart. This methodical approach—testing the same patient population, using the same assessment criteria, and obtaining consistent results across multiple sites—gave the FDA the confidence that LaViv actually worked for this specific condition.
The nasolabial fold was also an ideal area to test because it presents a defined anatomical space with visible, measurable wrinkles that don’t move as much as other facial features. When a patient smiles, the nasolabial fold deepens predictably, making before-and-after comparisons straightforward for trained evaluators. For scarring, the clinical picture is far more complex: acne scars vary dramatically in type (rolling, boxcar, atrophic, hypertrophic), depth, and location, which would require different study designs to demonstrate efficacy across multiple scar subtypes. The company that developed LaViv did not submit clinical data for scars, so the FDA had no basis for approving that indication.

The Clinical Trial Design That Led to FDA Approval
The two pivotal Phase III trials that supported LaViv’s approval were methodologically sophisticated. Each study was randomized, meaning patients were assigned by chance to either receive LaViv or placebo injections. Both studies were double-blind, so patients didn’t know whether their injections contained active cells or a control solution, and the physician-evaluators assessing wrinkle improvement were kept separate from the treating physicians administering the injections. This separation of roles prevented bias—evaluators couldn’t unconsciously judge results more favorably because they knew who got the real treatment.
The treatment protocol required three sessions conducted 3 to 6 weeks apart (approximately 5 weeks in the trials), with each injection containing up to 2 milliliters of LaViv cells. The primary efficacy measure, assessed at six months after the final treatment, looked for at least a two-point improvement on a standardized nine-point wrinkle severity scale. This six-month timeframe was important because it gave the injected cells time to proliferate, differentiate, and produce collagen in the dermal layer of skin. However, if you’re considering LaViv, keep in mind that efficacy can vary; not all patients in the trials achieved the two-point improvement, which is why informed expectations are essential before committing to the procedure.
How LaViv Differs from Dermal Fillers and Botox for Nasolabial Folds
Patients seeking treatment for nasolabial folds typically choose between three main approaches: temporary dermal fillers (hyaluronic acid, calcium hydroxylapatite, or poly-L-lactic acid), neurotoxins like botulinum toxin (Botox), or cellular therapies like LaViv. Dermal fillers provide immediate volume that plumps the skin, softening the appearance of nasolabial folds, but results typically last 6 to 18 months depending on the product, after which patients need repeat injections. Botox relaxes the muscles that deepen the fold when smiling, but effects are temporary and fade after 3 to 4 months.
LaViv takes a completely different biological approach: it doesn’t fill space or paralyze muscle; instead, it stimulates your own fibroblasts to produce new collagen within the skin itself. Because it uses your own cells, the body is less likely to reject or degrade them, and improvements can be more durable than fillers—though the exact duration of results varies among patients. The tradeoff is complexity: LaViv requires a minor skin biopsy behind the ear, a 2-3 week cell culturing process, and then three injection appointments spread over weeks, compared to the convenience of a single filler injection. This more involved process is why LaViv is typically recommended for patients seeking a longer-term solution rather than a quick fix.
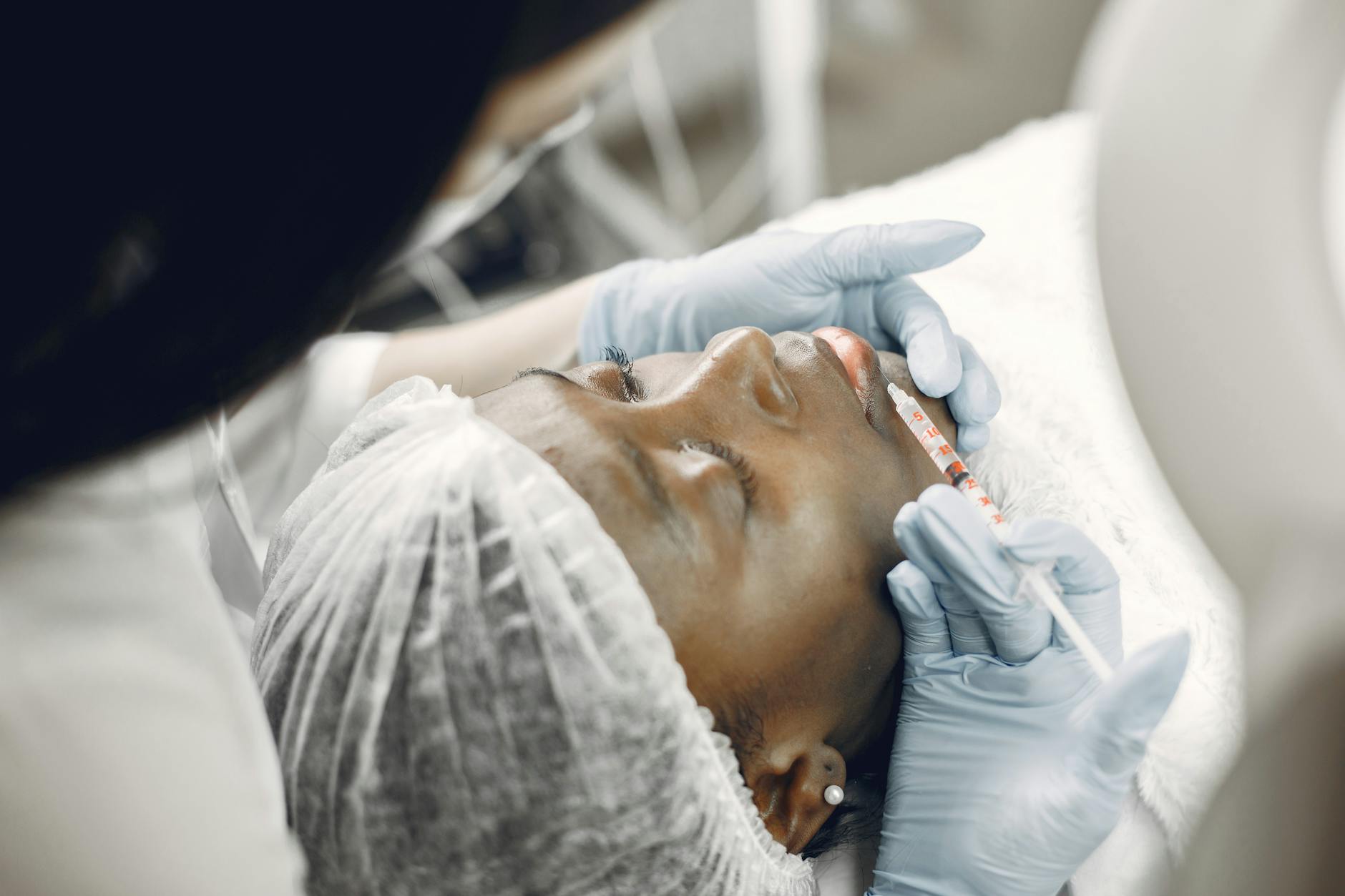
The LaViv Treatment Process: From Biopsy to Injection
The LaViv procedure begins not with an injection but with a minor surgical biopsy. A dermatologist or plastic surgeon removes a small piece of skin (approximately 2-3 millimeters) from behind the ear—an area called the post-auricular region—where scarring is hidden and healing is typically excellent. This biopsy site is closed with a single suture or left to heal on its own, depending on the size. The biopsied tissue is then shipped to the manufacturing facility, where your fibroblasts (skin cells) are carefully extracted, cultured, and expanded over 2 to 3 weeks until there are sufficient cells to fill the treatment vials. Once the cells are ready, you return for three treatment sessions spaced 5 weeks apart.
At each appointment, the physician injects up to 2 milliliters of your cultured fibroblasts directly into the dermis (deeper layer) of the nasolabial fold using a fine needle. The injection causes mild swelling and bruising that typically resolves within a few days. Between treatments, your newly injected cells are beginning to integrate into the skin and start producing collagen. By the time you receive your third injection, the cells from the first treatment have already been working for approximately 10 weeks. Results continue to improve over the 6 months following the final treatment as collagen remodeling progresses, which is why the FDA trials measured efficacy at the six-month mark rather than immediately after injections.
Why LaViv Wasn’t Approved for Acne Scars Despite Research Potential
While LaViv represents an innovative approach to skin rejuvenation, it received FDA approval only for nasolabial folds, not for acne scarring. This distinction is crucial because many patients and even some practitioners wonder whether the same cellular mechanism that works for wrinkles might also improve scars. Early research has explored this possibility, and the biological rationale is appealing: if LaViv stimulates collagen production, couldn’t it fill in atrophic acne scars by rebuilding lost tissue? However, research potential is not the same as regulatory approval, and the FDA requires specific clinical evidence before approving a new indication. The fundamental limitation is that acne scars are structurally and pathologically different from nasolabial wrinkles.
A nasolabial fold is primarily a dynamic wrinkle caused by repetitive muscle contractions and loss of dermal elasticity; it responds predictably to collagen augmentation. Acne scars, by contrast, are heterogeneous—rolling scars involve loss of dermal support and tissue attachment; boxcar scars are discrete, steep-sided depressions; atrophic scars are poorly defined indentations. Each subtype may respond differently to cellular therapy, and a single clinical trial protocol might not adequately demonstrate efficacy across all scar types. Without a dedicated, well-powered clinical trial specifically designed for acne scarring with clear efficacy endpoints, the FDA will not grant approval for that indication. Using LaViv off-label for scars is possible but remains unproven and is not recommended as a first-line treatment.

Clinical Outcomes and Safety Data from the Pivotal Trials
In the two Phase III trials, the efficacy results met the FDA’s primary endpoint: a statistically significant percentage of LaViv-treated patients achieved at least a two-point wrinkle improvement at six months post-final treatment compared to placebo. Beyond this primary measure, researchers also documented patient satisfaction, durability of results, and safety. Most treated patients reported visible improvement in the appearance of their nasolabial folds, and improvement continued beyond the six-month mark as ongoing collagen remodeling occurred. Some patients maintained benefit for well over a year, though individual results vary.
Regarding safety, the most common side effects were injection-site reactions: swelling, bruising, redness, and tenderness that resolved within days to weeks. Serious adverse events were rare, but they were documented and disclosed to the FDA during the approval process. The use of autologous cells (your own cells) is inherently safer than introducing foreign proteins or synthetic materials, because your immune system recognizes the cells as self and doesn’t attack them. However, the biopsy procedure itself carries small risks of infection and scarring, though these are uncommon when the procedure is performed by experienced providers. Anyone considering LaViv should discuss both the realistic benefits and potential risks with their treating physician.
The Future of Cellular Therapies in Aesthetics and Where LaViv Stands
LaViv’s FDA approval in 2011 was groundbreaking—it represented the first aesthetic cellular therapy approved by the agency and opened a regulatory pathway that other companies have since explored. Since then, the field of regenerative aesthetic medicine has grown substantially, with research into stem cell therapies, platelet-rich plasma, and other cellular approaches expanding rapidly. However, LaViv remains the only FDA-approved personalized autologous fibroblast therapy for any aesthetic indication, which speaks to both its uniqueness and the high bar the FDA sets for cellular therapeutic claims.
Looking forward, the question of whether LaViv or similar cellular therapies will gain approval for additional indications—including acne scars—depends on whether companies invest in properly designed clinical trials for those conditions. The biologic mechanism is sound: stimulating fibroblasts to produce collagen is a rational approach to scar treatment. But biology alone doesn’t drive FDA approval; rigorous clinical evidence does. For now, patients with acne scars seeking cellular or regenerative approaches should be aware that proven, FDA-approved options for scars remain limited, and they should discuss what evidence exists for any proposed treatment before proceeding.
Conclusion
LaViv was FDA-approved on June 21, 2011, for moderate to severe nasolabial fold wrinkles because the company submitted high-quality clinical evidence from two Phase III randomized, double-blind, placebo-controlled trials involving 421 patients. These trials demonstrated that the treatment could achieve meaningful wrinkle improvement at six months post-treatment through a novel mechanism: injecting patients’ own cultured fibroblasts to stimulate natural collagen production. This autologous cellular approach differs fundamentally from temporary fillers or neurotoxins, offering the potential for more durable results—though it requires more time and effort from both patient and provider.
If you’re considering LaViv or exploring options for nasolabial folds, consult with a board-certified dermatologist or plastic surgeon who is trained in administering the treatment and can discuss realistic expectations, the complete treatment timeline, and whether this approach is right for your goals. For acne scars, recognize that while early research into cellular therapies is promising, LaViv is not FDA-approved for that indication, and you should evaluate other proven scar treatments in consultation with a specialist. The regulatory approval of LaViv demonstrates what rigorous clinical evidence can achieve, and it remains a meaningful option for patients seeking a longer-term approach to facial rejuvenation.
You Might Also Like
- Why Poly-L-Lactic Acid Differs from Hyaluronic Acid for Scars
- Why PRF Has Higher Growth Factor Concentration Than PRP for Scars
- Why Growth Factor Concentrates Outperform Standalone PRP for Scars
Browse more: Acne | Acne Scars | Adults | Back | Blackheads